HP 50g / 49g+ / 48gII graphing calculator
Advanced user’s reference manual
Page
Acknowledgements
Printing History
Page
Contents
Alog
Names
Algb
Amort
317
→COL
Delalarm
Delay
COL→
359
Expan
Findalarm
EXP2POW
Expand
IDIV2
Halt
IBP
Iferr
List
MIN
MAX
Maxr
Not
Parity
PA2B2
Parametric
Parsurface
3183
Roll
RND
Rnrm
Rolld
3226
Tail
Tabvar
→TAG
TAN
Utpc
Wireframe
Updir
Utpf
Contents
514
→ALG Apeek ARM→ ASM ASM→
610
Toff Tpar
Alrmdat
CST Exited Expr Iopar MASD.INI
Vpar
Contents of a Program
Understanding Programming
Examples of Program Actions
Calculations in a Program
Program Results
To enter a program
To enter commands and other objects in a program
Entering and Executing Programs
To store or name a program
3Q!Ì*4#3/* @Ë@Ï
To stop an executing program
3Q!Ì*4`3/*@Ï
@% @Ér # O4 /3 *!Ì Q3 @Ë@Ï
Program Keys Comments
Level
Osph K
To view or edit a program
To switch between entry modes
Viewing and Editing Programs
@%SPH% ˜
Creating Local Variables
Using Local Variables
Creating Programs on a Computer
`J!%SPH% @%SPH% ˜
Program Comments
`OSPHLV K
Evaluating Local Names
Defining the Scope of Local Variables
Compiled Local Variables
Creating UserDefined Functions as Programs
Key Programmable Description Command
Using Tests and Conditional Structures
Testing Conditions
To include a test in a program
Logical Functions
Using Logical Functions
Keys Programmable Description Command
TEST% L
Press !%BRCH% !%IF%
Using Conditional Structures and Commands
To enter if then END in a program
To enter IFT in a program
To enter Ifte in a program or in an algebraic
To enter if then Else END in a program
Press !%BRCH% @%#IF#%
Press !%BRCH% L!%IFTE%
To enter Case END in a program
Conditional Examples
Otst K
26 `52 J %TST%
Using Definite Loop Structures
Using Loop Structures
While … Repeat … END
To enter Start Next in a program
Start Next Structure
« … start finish Start loopclause Next … »
Press !%BRCH% !%START%
To enter Start Step in a program
Start Step Structure
« … start finish Start loopclause increment Step … »
Press ! %BRCH% … %START%
To enter for Next in a program
For Next Structure
« … start finish for counter loopclause Next … »
Press !%BRCH% ! %FOR%
To enter for Step in a program
For Step Structure
« … start finish for counter loopclause increment Step … »
Press ! %BRCH% … %FOR%
« … do loopclause Until testclause END … »
Using Indefinite Loop Structures
Do Until END Structure
To enter do Until END in a program
Duplicates n, stores the value into n
To enter While Repeat END in a program
While Repeat END Structure
« … While testclause Repeat loopclause END … »
Press !%BRCH% ! %WHILE%
To enter Incr or Decr in a program
Using Loop Counters
Press ! #MEM# %ARITH% %INCR% or %DECR%
Using Summations Instead of Loops
∑ j
Types of Flags
Using Flags
Setting, Clearing, and Testing Flags
To set, clear, or test a flag
Month/day/year format
Recalling and Storing the Flag States
Example
Day.month.year format
Execute Rclf !L %MODES% %FLAG% L%RCLF%
Using Subroutines
To recall the current flag states
To change the current flag states
Torsv K
Torsa K
` 8 J %TORSV%
Press !LL %RUN% %KILL%
To singlestep from the start of a program
To turn off the Halt annunciator at any time
@·J6 `8 `O%TORSV% !LL%RUN% %DBUG%
To singlestep from the middle of a program
To singlestep when the next step is a subroutine
@·J10 `12 O%TORSV% !LL%RUN% %DBUG%
To cause a userdefined error to occur in a program
Trapping Errors
Causing and Analyzing Errors
SingleStep Operations
Error Trapping Commands
To artificially cause a builtin error to occur in a program
To analyze an error in a program
%ERROR%
Press !LL %ERROR% !%IFERR%
Making an Error Trap
« … Iferr trapclause then errorclause END … »
Iferr then END Structure
Iferr then Else END Structure
Press !LL %ERROR% @%IFERR%
To enter Iferr then Else END in a program
Key Command Description
Data Input Commands
Using PROMPT, Cont for Input
Input
`OTPROMPT ‰
@·J %TPROM%
To enter Disp Freeze Halt in a program
Using Disp Freeze HALT, Cont for Input
To respond to Halt while running a program
To enter Input in a program
Using INPUT, Enter for Input
« … promptstring commandline Input OBJ→ … »
To respond to Input while running a program
%VSPH%
To include Input options
Commandline cursorposition operatingoptions
To design the commandline string for Input
Cursorposition
Press @ Õ ! Ê a @
To process the result string from Input
`O Tinput ‰
%TINPU%
Using Inform and Choose for Input
To set up an input form
To set up a choose box
`OPHONES ‰
To enter Beep in a program
Using Wait for Keystroke Input
Beeping to Get Attention
To enter Wait in a program
Output
Using KEY for Keystroke Input
Data Output Commands
OUT%
To label a result with a tag
Labeling Output with Tags
Labeling and Displaying Output as Strings
%TTAG% 1.5 ˜1.85 `
To set up a message box
Pausing to Display Output
Using Msgbox to Display Output
`OTSTRING ‰
Using Menus with Programs
Using Menus for Input
Olst ‰
Using Menus to Run Programs
To create a menubased application
SI%
@·J%EIZ%
@%%I%% 2*%%I%% !%%E%%
Turning Off the Calculator from a Program
%% !Ü.37~@6 68 %%I%% !%%Z%%
To turn off the calculator in a program
Page
FIB1 Fibonacci Numbers, Recursive Version
Fibonacci Numbers
RPL Programming Examples
%FIB1%
FIB2 Fibonacci Numbers, Loop Version
FIB1% !Ü10 N
%FIB2%
FIB2 program listing
10 %FIB2%
Structured programming. Fibt calls both FIB1 and FIB2
Fibt Comparing ProgramExecution Time
Techniques used in Fibt
Required Programs
13 %FIBT%
Displaying a Binary Integer
PAD Pad with Leading Spaces
Techniques used in PAD
`OPAD K
Preserve Save and Restore Previous Status
PAD program listing
Techniques used in Preserve
Preserve program listing
Bdisp Binary Display
`OPRESERVE K
Techniques used in Bdisp
PAD
Bdisp program listing
`OBDISP K
Tile Percentile of a list
Median of Statistics Data
144 %BDISP%
Tile program listing
Median Median of Statistics Data
Techniques used in %TILE
`O%TILE K
Techniques used in Median
Required Program
18 `12 `˜šš
Expanding and Collecting Completely
`OMEDIAN K
11 `1 `
Multi program listing
Multi Multiple Execution
Techniques used in Multi
`OMULTI K
Exco program listing
Exco Expand and Collect Completely
Techniques used in Exco
`OEXCO K
Ü4 *Y+Z +
Minimum and Maximum Array Elements
MNX Minimum or Maximum Element-Version
User and system flags for logic control
MNX program listing
`OMNX K
12 `56 `˜šš 45 `1 `
MNX2 Minimum or Maximum Element-Version
Techniques used in MNX2
MNX2 program listing
Techniques used in Aply
Applying a Program to an Array
%MNX2%
Aply program listing
Make sure the flag 1 is clear to begin
Ô3†2W†4` ‚Å 3 QA *7 `J %APLY% H#DISP ˜˜
Converting Between Number Bases
`OAPLY K
Techniques used in nBASE
NBASE program listing
Verifying Program Arguments
1000 `23 J %NBASE%
Names program listing
Names Check List for Exactly Two Names
Techniques used in Names
`ONAMES K
Techniques used in VFY
VFY Verify Program Argument
VFY program listing
Ojeff ` Osarah `
Converting Procedures from Algebraic to RPN
Oben `
%LIST% %²LIST%
Techniques used in →RPN
→RPN program listing
XQ3 `%²RPN%
Bessel Functions
Techniques used in BER
%BER%
BER program listing
`OBER K
%BER% !Ü2
Techniques used in Sintp
Animation of Successive Taylor’s Polynomials
Sintp Converting a Plot to a Graphics Object
Sintp program listing
Setts program listing
Setts Superimposing Taylor’s polynomials
Techniques used in Setts
`OSETTS K
TSA program listing
TSA Animating Taylor’s Polynomials
Techniques used in TSA
`OTSA K
Techniques used in PIE
Programmatic use of matrices and statistics commands
Programmatic Use of Statistics and Plotting
Temporary menu for data input
Angle
`OPIE K
983 %SLICE% 416 %SLICE% 85 %SLICE%
ΑENTER program listing
Trace Mode
Techniques used in αENTER and ßENTER
ßENTER program listing
Rootr program listing
InverseFunction Solver
Techniques used in Rootr
`OROOTR K
%X²FX% ` 599.5 ` 1 %ROOTR%
Animating a Graphical Image
@Å@É x †O3.7
Techniques used in Walk
`OWALK K
Type
Introduction
Acos
Input/Output
Other Provided Details
How Commands Are Alphabetized
Parallel Processing with Lists
Computer Algebra System Commands and Functions
Classification of Operations
Library identifier
Backup identifier
Realnumber time or angle in hoursminutessecond s format
Binary integer
ABS
Abcuv
ACK
Ackall
Type Description
ACOS2S
ASIN, ATAN, COS, Isol
Acosh z
Acosh
ASIN2C, ASIN2T, ATAN2S
Symb ACOSHsymb
ADD
Addtmod
Alog
Addtoreal
Algb
10z
Principal Interest Balance
Amort
TVM, TVMBEG, TVMEND, Tvmroot
Animate
ANS
Apply
Access …µAPPLY Input/Output
ARC
Archive
ARG
ARRY→
Access …µARRY→ Input/Output
Arit
→ARRY
Access !¼ ¼is the leftshift of the Skey
ASIN2T
ASIN2C
Asinh
Symb ASINHsymb
ASN
Asinh z
ACOSH, ATANH, ISOL, Sinh
Assume
Access … ÃL BIT ASR
ASR
Skey
Atan
ADDTOREAL, Unassume
Symb ATANsymb
ATAN2S
Atan z
ACOS, ASIN, ISOL, TAN
Atick
Access …µATICK
Atanh
Atanh z
Augment
Access …µATTACH Input/Output
Attach
#n #m
Axes
Access …µAUTO Input/Output None
Auto
BAR
AXM
Access …µAXES Input/Output
AXL
Atick xaxis label yaxis label
AXL, AXQ
AXQ
BAR
AXL, AXM, GAUSS, QXA
Basis
Access …µBAR Input/Output None
Barplot
Baud
Beep
Access …µBESTFIT Input/Output None
Access … Ã BIN
Bestfit
Blank
Access !L Pict BOX Is the leftshift of the Nkey
Bins
BOX
Bytes
Access …µBUFLEN Input/Output
Buflen
#n1 #m1 #n2 #m2 X1, y1 X2, y2
C2P
Access … Ã B → R
Cascfg
Case
Cascmd
Help
Xunit Nunit Symb CEILsymb
Ceil
Centr
FLOOR, IP, RND, Trnc
Chinrem
Cholesky
Choose
Command Result
CHR
NUM, POS
Circ
Cksm
REPL, SIZE, SUB
Cllcd
Clear
Clkadj
Closeio
Clvar
CLΣ
Clusr
Cmplx
COL→
→COL
COL
Collect
COL+
Colct
Symb1 Symb2
CON
Colσ
Comb
Symb COMBsymb ,m COMBn, symb COMBsymb ,symb
Carray constant Name
Cond
Rarray constant
IDN
Conic
Access …µCONIC Input/Output None
Conj
Constants
Conlib
Const
Rarray Carray Symb CONJsymb
Corr
Cont
Convert
X1unitssource X2unitstarget X3unitstarget
Cosh
Access Flags
COS
COV
Cswp
Crdir
Cross
…µCR
Cylin
Curl
Cyclotomic
COL+, COL-, Rswp
Date
→PX
Darcy
#n, #m
Dbug
→DATE
DATE+
→date
Decr
Ddays
DEC
« program » or program name
Define
Dedicace
DEF
Result See also
Degree
Delalarm
DEG
Name=exp Namename Name =expname
Delkeys
Delay
RCLALARM, Stoalarm
CR, OLDPRT, PRLCD, PRST, PRSTC, PRVAR, PR1
Depth
Access …µDEPND Input/Output
Depnd
Xkey1, ... ,xkey n
Dervx
Deriv
Desolve
Detach
Access …µDETACH Input/Output
DET
Attach
→DIAG
DIAG→
Diagmap
Diff
Diffeq
DIR
Access …µDIFFEQ Input/Output
Disp
Obj List
Dispxy
Distrib
FREEZE, HALT, INPUT, Prompt
DIV2MOD
DIV
DIV2
CURL, Hess
Divpc
Divis
Divmod
DIV2
Until testclause END
TAYLOR0, TAYLR, Series
DERIV, DERVX, DESOLVE, ∂
END
Doerr
Error
Dolist
Dosubs
List List n « program » Results Command Name List n+1
Domain
SIGNTAB, Tabvar
DOLIST, ENDSUB, NSUB, Stream
List « program » Command Name
DOT
CNRM, CROSS, DET, Rnrm
Draw
Access …µDRAW
Access …µDRAX Input/Output None
DRAW3DMATRIX
DROP2
Droite
Drop
Lagrange
DUP
Dropn
Dtag
DUP2
Dupdup
Dupn
Editb
Edit
Egcd
Else
EGV
Egvl
END
EPSX0
Endsub
ENG
Sign mantissa E sign exponent
EQ→
Eqnlib
EQW
Erase
Errn
ERR0
Errm
Euler
Obj See above
Eval
Exlr
→NUM
EXP
EXP&LN
Ex,y = excosy + iexsiny
EXP2POW
EXP2HYP
Expan
COLCT, EXPAND, ISOL, QUAD, Show
Expand
Expandmod
Result X2+X
Expm
Expfit
Expln
Eyept
Factor
F0λ
Fact
…µF0λ
Factors
Factormod
Modsto
Fanning
Access …µFANNING
FAST3D
Fcoef
Access …µFAST3D Input/Output None
Access ! Test LL FC ? C
FC?
CF, FC?, FS? FS?C, SF
Fdistrib
FFT
Distrib
Filer
Findalarm
Access …µFINISH
Finish
FONT6
Flasheval
Floor
Xunit Nunit Symb FLOORsymb
FONT→
FONT7
FONT8
→FONT
For
Freeze
Fourier
Free
Xunit Yunit Symb FPsymb
Display Area Value Code
Froots
FS?
CLLCD, DISP, Halt
Function
FS?C
CF, FC?, FC?C, FS?C, SF
Fxnd
Access …µFUNCTION
Exlr
Gbasis
Gamma
Gauss
AXQ, QXA
GET
GCD
Gcdmod
GCDMOD, EGCD, IEGCD, LCM
Objget
Geti
GETI, PUT, Puti
GET, PUT, Puti
GOR
Grad
#n #m Grob1
Greduce
Gramschmidt
Graph
DEG, RAD
Gridmap
Access …µGRIDMAP Input/Output None
Access …µ→GROB
→GROB
Gxor
Grob
Grobadd
→LCD, LCD→
Halt
Hadamard
Halftan
GOR, REPL, SUB
→HEADER
Head
HEADER→
Help
HEX
Hermite
Hess
CURL, DIV
Hilbert
Histogram
HMS
Access …µHISTPLOT Input/Output None
Histplot
HMS+
… & 9L HMS +
HMS→
→HMS
… &9L HMS →
Horner
Home
Iabcuv
IBP
Ibasis
Ibernoulli
ABCUV, Iegcd
Results See also
Ichinrem
IDN
Chinrem
DIV2, Iquot
IDIV2
Iegcd
Ax+by=c
Case
Iferr
ELSE, END, IFERR, then
∑ Y k e
Ifft
Ilap
IFT
Ifte
Obj It depends
…ßIM
Image
LAP, Lapl
Rarray Carray Symb IMsymb
Inform
Incr
Indep
Global Global xstart xend
Input
Resets
Title Format Resets Init → vals
Init
INT
Stack prompt Commandline prompt Result
PROMPT, STR→
INV
Integer
Intvx
INTVX, Risch
Invmod
Command INVMOD2 Result
Iquot
Isom
Iremainder
Isol
Symb1 Global Symb2
Mkisom
ISPRIME?
Jordan
NEXTPRIME, Prevprime
KEY
KER
Kerrm
BASIS, Image
Keyeval
Access …µKEYEVAL Input/Output
→KEYTIME
Kill
KEYTIME→
Kget
Label
LANGUAGE→
Access …µ→LANGUAGE Input/Output
Lagrange
→LANGUAGE
Last
LAP
Lapl
Objn Obj1
→LCD
Lastarg
LCD→
LCM
Lcxm
Ldec
Libs
Lgcd
Libeval
HERMITE, Tchebycheff
LIN
Lim
Limit
Title, nlib, nport, ...,title, nlib, nport
Linfit
Line
Σline
Model Form of Expression
LIST→
Linin
Linsolve
→LIST
Πlist
List
Σlist
Access …¹ ¹is the rightshift of the Qkey
Lncollect
Lname
LNP1
Ln x +
Local
LOG
Symb LNP1symb
Access !Ø Input/Output
Logfit
LSQ
Access …µLR Input/Output
Full Command and Function Reference 3139
Intercept x Slope x
DET, INV
Lvar
MAD
Lname
Mant
Access …µMAP Input/Output
Main
MAP
↑MATCH
Access …µ↓MATCH Input/Output
Access …µ↑MATCH
Symb Symb symb
MAX
Maths
Matr
Symb Symb , symb
Mcalc
Maxr
Maxσ
Input/Output See also
Menu
Mean
MEM
BINS, MAXΣ, MINΣ, SDEV, TOT, VAR
Menuxy
RCLMENU, Tmenu
Minehunt
Merge
MIN
MENU, Tmenu
Minit
MINIFONT→
→MINIFONT
Minr
Mkisom
Minσ
Mitm
00000000000E-499
Modular
MOD
Modsto
Mod y
Msgbox
Molwt
Mroot
Name Or xunit String
Mslv
Msolvr
→NDISP
Multmod
Muser
EQNLIB, MCALC, MINIT, MITM, MROOT, MSLV, Muser
Ndupn
Access …ß NEG ßis the rightshift of the 1key
Ndist
NEG
Next
Newob
Nextprime
Not
Access ! Test L not Is the leftshift of the Nkey
NIP
Noval
NUM
Nsub
→NUM
…µNΣ
Numy
Numx
OBJ→
Oldprt
OCT
OFF
… &NL OBJ →
Access …µOPENIO
Access …µOLDPRT
Openio
Memory Directory Lorder is the leftshift of the Nkey
Order
Over
Global1 ... globaln
Parametric
P2C
PA2B2
Xmin, ymin, xmax, ymax, indep, res, axes, ptype, depend
Access …µPARITY Input/Output
Parity
Parsurface
Partfrac
Pcoef
Path
Pcar
Home directoryname 1 ... directoryname n
Pcontour
Access …µPCONTOUR Input/Output None
Pcov
Perm
Pdim
Perinfo
Xmax, ymax
Pgdir
Pertbl
Peval
Pick
Picture
Pict Command Puts the name Pict on the stack
Pict
Pinit
Pixoff
Access …µPKT
PIX?
Pixon
Pmax
Plot
Plotadd
Pmin
Finds the minimal polynomial of a matrix
Pmini
Polar
An nxn matrix a
POP
Access …µPOLAR
Polynomial
Form of Current Plotting Action Equation
Potential
Input An expression raised to a power
POS
Powexpand
Powmod
PR1
…µPR1
Predv
Predx
Object
Predy
Preval
Prompt
Prevprime
Prlcd
Promptsto
Global→
Proot
Propfrac
Prompt STO
Prvar
Prst
Prstc
Name Name1 name2 Nport global
Psi
Psdev
PSI
MEAN, PCOV, PVAR, SDEV, TOT, VAR
Purge
Ptayl
Ptprop
Symb String or x or xunit or name
Global Global1 ... globaln
Push
PUT
CLEAR, CLVAR, NEWOB, Pgdir
Obj put List
This command sequence
Puti
Obj put List Name
Pvars
Pvar
MEAN, PCOV, PSDEV, SDEV, VAR
Pview
Access …µPVARS Input/Output
Nport namebackup Memory
Pwrfit
→Qπ
PX→C
+ c/d*i
+ c/ d
Quad
̟ + c/ d* ̟
LQ, LSQ
Arithmetic, !ÞPOLYNOMIAL !«
Quot
Quote
Quotient of the Euclidean division
Rand
QXA
RAD
Rank
Ratio
Access …µRATIO
Ranm
LQ, LSQ, QR
Rcij
Rceq
RCI
Steq
Rclalarm
RCL
Rclkeys
FINDALARM, Stoalarm
Rclf
Rclmenu
Rcws
Rclvx
Rclσ
RDM
COMB, PERM, Rand
RDZ
…ßL RE
Recv
Recn
Rect
REsymb
Quot
Remainder
Rename
New name Old name
Repl
Reorder
Repeat
END, While
String target
Plot Type Default Interval
RES
String String result
Resultant
Restore
Backup To restore a
Slopefield
Rewrite
Revlist
Risch
RKF
Rkferr
Rkfstep
Problem for a differential equation
Is the rightshift 3key
RLB, RR, RRB
RLB
RND
RNDsymb
Rolld
Rnrm
Roll
Romupload
ROW
Root
ROT
3207
ROW→
ROW+
→ROW
RRB
Access … ÃL Byte RRB Is the rightshift of the 3key
RPL
RL, RLB, RRB
Rref
Rref
Rrefmod
RRK
− 2 t + t 2
Rrkstep
RSD
Rsberr
RKF, RKFERR, RKFSTEP, RRK, Rsberr
Rules
Access … Ã R → B
Rswp
DET, IDN
180/̟x
Same
→R, IM, RE
→Dsymb
Scale
Access …µSCALEH Input/Output
Sbrk
Scaleh
Scatter
Scatrplot
AUTO, SCALEH, Xrng
SCI
Access …µSCATTER Input/Output None
Schur
Sclσ
Sdev
Sconj
Scroll
Send
Series
SEQ
DOSUBS, Stream
Server
Seval
Sigma
Show
Sidens
X1/cm3
Sigmavx
Access !ÖDERIV L
Access !ÖDERIV LL
Sign
SIGNsymb
Signtab
SIMP2
ABS, MANT, Xpon
Sincos
Simplify
SIN
Sin z
Size
Sinh
Sinv
Sinh z
String Integer List Vector
SLB
Slopefield
Symb Grob
Snrm
Sneg
NEG, SCONJ, Sinv
Solve
Solveqn
Solvevx
Solver
Sort
Square Analytic Function Returns the square of the argument
Sphere
Srad
SQsymb
COND, SNRM, Trace
SRB
Srecv
ASR, SL, SLB, SR
SST
Access …µSRECV
Srepl
SST↓
STD
Start
Step
Steq
Access …µSTIME
Step
Stime
STO
Stoalarm
Access Input/Output
Buflen
Stokeys
Access …µSTOF
Stof
Óis the rightshift of the 9 key
Stovx
Store
STO+
STO/, +
→STR
Stoσ
STR→
DTAG, EQ→, LIST→, OBJ→, →STR
Sturm
Stream
Strm
List Obj Result
SUB
Sturmab
Stws
Sturmab
X1, y1 X2, y2
Subst
Subtmod
CHR, GOR, GXOR, NUM, POS, REPL, Size
Swap
SVD
SVL
DIAG→, MIN, SVL
Sylvester
Syseval
SYST2MAT
X, symb
Tabval
100 y
Symb 1, symb
Tail
Tabvar
→TAG
→ARRY
TAN2SC
TAN
TAN2CS2
Tan z
Tanh z
TAN2SC2
Tanh
TANHsymb
Tchebycheff
TAYLOR0
Taylr
HERMITE, Legendre
Tests
Tcollect
Tdelta
TDELTAsymb1, symb2
Text
Teval
Texpand
Then
→TIME
Ticks
Time
Tinc
TINCsymb1, symb2
Tlin
Tline
SIMPLIFY, TCOLLECT, Texpand
Trace
Tmenu
TOT
Tran
Effect
Transio
Trig
BAUD, CKSM, Parity
Trigsin
Trigcos
Trigo
Trigtan
Trnc
TRN
CONJ, Tran
TRNCz
Trunc
Truth
TRNCsymb
Tsimp
Access …µTRUTH Input/Output None
Tstr
Tvmbeg
Tvars
TVM
Tvmend
Ubase
Builtin Commands
Type
Object Type Number User objects
Unassign
Ufact
UFL1→MINIF
Unassume
→UNIT
Unbind
Unpick
Updir
Unrot
Until
Utpc
UTPF, UTPN, Utpt
Utpf
Utpn
UTPC, UTPN, Utpt
Uval
Utpt
UVALsymb
X1, x2
Flag -16 clear Rectangular mode Flag -16 set Polar mode € y
Coordinate System -16, Complex Mode
X1, x2, ..., xn Xn-2
Vandermonde
VAR
VER
Access …µVERSION Input/Output
Vars
Version
Visit
Variable name Contents opened in the command line Editor
Variable name Contents opened in the most suitable Editor
Visitb
Vtype
Access ! L in Wait Is the leftshift of the Nkey
Wait
While testclause Repeat loopclause END
Wireframe
While
While Repeat
Wslog
ΣX2
Access …µWSLOG
Access …µΣX Input/Output
Log4 ... log1
Xget
ΣX2
Xcol
Xmit
Xnum
Access …µXMIT
XOR
XPONsymb
Xpon
Xput
MANT, Sign
Xrecv
Command XQ.3658 Results 1829/5000
Command XQ.3658 Results √19/142
Xrng
Xserv
Access …µXSEND
Xsend
Input/Output RPN
ΣXY
Xvol
Xxrng
ΣX*Y
Ycol
ΣY2
ΣY2
…µΣY
Yslice
Yrng
AUTO, PDIM, PMAX, PMIN, Xrng
Zeros
Yvol
Yyrng
EYEPT, XVOL, XXRNG, YYRNG, Zvol
Zfactor
Power
Access Q
Zvol
Zsymb
Where
√ Square Root
Symbz
SQ, , Isol
? Undefined
Access …Á Áis the rightshift of the Ukey
Integrate
Lower limit, upper limit, integrand, name
Sigma Plus
∞ Infinity
Summation
Results -π/2
Sigma Minus
Access …µΣ+ Input/Output
Access …µΣ Input/Output
X 2, …, x m 1, …, x 1 m x n 1, … , x n m
14159265359…
∂ Derivative
Factorial
Symb Name Xunit
Xy/100
Percent
Unit attachment
Symb,x
Less than
« » Program delimiters
Type Object Description
≤ Less than or Equal
#n1 #n2
Greater than
≥ Greater than or Equal
≠ Not equal
Multiply
≠ symb
+ Add
Subtract
Divide
= Equal
Y1/unit
== Logical Equality
DEFINE, RCL, →, STO
Store
→ Create Local
… É
Semicolon
CLEAR, DROP, DROPN, DROP2
Points to note when choosing settings
CAS Settings
Selecting CAS Settings
CAS directory, Casdir
Computer Algebra System
Compatibility with Other Calculators
Using the CAS
Examples and Help
Extending the CAS
Algebra commands, …×
Computer algebra command categories listed by menu
Arithmetic commands
Arithmetic Integer commands, !ÞINTEGER
Arithmetic Permutation commands, !Þ Permutation
Arithmetic Modulo commands, !Þ Modulo
Other Arithmetic commands, !Þ
Calculus commands
Create, !Ø Create
Exp and Lin commands, !Ð
Matrixrelated commands
Operations, !Ø Operations
Linear Applications, !Ø Linear Appl
Quadratic form, !Ø Quadratic Form
Linear Systems, !Ø Linear Systems
Eigenvectors, !Ø Eigenvectors
Other Trigonometry commands, …Ñ
Symbolic solve commands, !Î
Trigonometry commands
Hyperbolic, …Ñ Hyperbolic
Base conversion tools, !Ú Base
Convert commands, !Ú
Unit conversion tools, !Ú Units Tools
Trigonometric conversions, !Ú Trig Conv
Other CAS operations, …µ
Matrix convert, !Ú Matrix Convert
These commands display menus or lists of CAS operations
CAS menu commands, …µ
CAS utility operations
Equation Reference
Equation Reference
FLUIDS, 24 *********************3
Σcr Critical stress Σmax Maximum stress
Columns and Beams
Variable Description
Internal bending moment at
Equations
Elastic Buckling 1
Eccentric Columns 1
Example
Simple Slope 1
Simple Deflection 1
Equation
Simple Moment 1
Solution V=624.387lbf
Simple Shear 1
Cantilever Slope 1
Cantilever Deflection 1
Cantilever Moment 1
Solution V=200lbf Equation Reference
Cantilever Shear 1
Relative permittivity
Electricity
Relative permeability
Ohm’s Law and Power 2
Coulomb’s Law 2
Solution I1=5.6250A
Wire Resistance 2
Solution V1=80V
Voltage Divider 2
Series and Parallel R 2
Solution R=0.175
Series and Parallel C 2
Capacitive Energy 2
Solution V=50V
Series and Parallel L 2
Inductive Energy 2
DC Capacitor Current 2
RLC Current Delay 2
Solution q=0.0020C Equation Reference
Capacitor Charge 2
RC Transient 2
Solution V=3.2968V
DC Inductor Voltage 2
RL Transient 2
Plate Capacitor 2
Solution I=0.0072A
Resonant Frequency 2
Cylindrical Capacitor 2,19
Solenoid Inductance 2
Toroid Inductance 2
Sinusoidal Voltage 2
Fluids
Sinusoidal Current 2
Reynolds number
Pressure at Depth 3
Bernoulli Equation 3
Initial and final velocities
Flow with Losses 3
Forces and Energy
Flow in Full Pipes 3
Linear Mechanics 4
Centripetal Force 4
Angular Mechanics 4
Hooke’s Law 4
Law of Gravitation 4
1D Elastic Collisions 4
Drag Force 4
MassEnergy Relation 4
Gases
Ideal Gas Law 5
Isothermal Expansion 5
Solution Vf=21
Ideal Gas State Change 5
Polytropic Processes 5
Real Gas Law 5
Real Gas State Change 5
Heat Transfer
Kinetic Theory 5
Thermal Expansion 6
Heat Capacity 6
Conduction 6
Convection 6
Conduction + Convection 6
Fba
Magnetism
Black Body Radiation 6
Ia, Ib
Straight Wire 7
Force between Wires 7
Magnetic B Field in Solenoid 7
Solution B=0.0785T
Magnetic B Field in Toroid 7
Motion
Object in Free Fall 8
Linear Motion 8
Projectile Motion 8
Terminal Velocity 8
Angular Motion 8
Circular Motion 8
Escape Velocity 8
Optics
Law of Refraction 9
Critical Angle 9
Brewster’s Law 9
Spherical Refraction 9
Solution n2=1.5000
Spherical Reflection 9
Thin Lens 9
Oscillations
Simple Pendulum 10
MassSpring System 10
Example Given L=15cm
Torsional Pendulum 10
Conical Pendulum 10
Simple Harmonic 10
Plane Geometry
Ellipse 11
Circle 11
Equations Example
Rectangle 11
Regular Polygon 11
Circular Ring 11
Triangle 11
Solid Geometry
Cone 12
Cylinder 12
Parallelepiped 12
Sphere 12
Solid State Devices
Drawn gate length Nmos Transistors, or
Saturation current density
Drawn mask length PN Step Junctions, or
Channel length JFETs
PN Step Junctions 13
Nmos Transistors 13
VDS
Bipolar Transistors 13
JFETs 13
Xdmax = ⋅ Vbi VGS + VDS
Maximum principal normal stress
Stress Analysis
Minimum principal normal stress
Shear Stress 14
Normal Stress 14
Stress on an Element 14
Mohr’s Circle 14
Example
Transverse Waves 15,1
Waves
Longitudinal Waves 15
Sound Waves 15
References
Page
Development Library
Obj1 … objn Symb Obj1, ...,objn
Development Library Command Reference
Obj String
Development Library
String Obj
BetaTesting
String Obj Debug string Error list
Code String
Library Object String
String Error list Edited string
Library n
LC~C
Ey,z.Et
→LST
Obj … obj Obj 1, ...,obj n
Pokearm
Obj 1 … obj n Obj 1, ...,obj n
Returns
→S2
String1 String2
String Global
Crlib Create Library Command
Xlib
Extension program
Starting Masd
Masd The Machine Language and System RPL Compiler
Introduction
Modes
Format of the error list
Errors
Error messages Message Description
Links
Labels
Extable
Constants
Expressions
Operator Priority
Macros and includes
Compilation directive
Filename conventions
Directive Description
!DBGINF directive
Docstr
CPU architecture
Saturn ASM mode
Bit registers
New instructions
Other notes
Skips
Skips instructions Equivalents
These instructions Are equivalent to
Tests
Saturn instructions syntax
Syntax Example
Syntax Example
DReg=hh
See above. This is only valid in emulated Saturn
Places the value of Exp in the code, on x nibbles
CARRY0
003AA Autousertest
ARM mode
ARM architecture
Instruction set
Description Form
Operation Assembler Action Flags
REG a
Armsat instruction
Offset Element
REG B
REG C REG D REG R0 REG R1 REG R2 REG R3 REG R4
Reals and system binary
System RPL mode
Instructions
Unnamed local variables
Tokens
Defines
Token Description
Code
LC 001 Gosbvl Outcinrtn ?CBIT=1.6
Turnmenuoff
LC 80100 Armsat
Disassemblers
Level
Nop
Entry Point Library Extable
Library
String String 1, ...,string n
Message Meaning # hex
Error and Status Messages A1
Messages Listed Alphabetically
C0C
A2 Error and Status Messages
Error and Status Messages A3
A4 Error and Status Messages
Σdat
Error and Status Messages A5
A6 Error and Status Messages
C0B
Error and Status Messages A7
C0E
C0D
A8 Error and Status Messages
Error and Status Messages A9
# hex Message General Messages
A10 Error and Status Messages
# hex Message
Error and Status Messages A11
OutofMemory Prompts
Object Editing Messages
Stack Errors and Messages
A12 Error and Status Messages
Equation Writer Application Messages
Array Messages
# hex Message FloatingPoint Errors
Error and Status Messages A13
Statistics Messages
Mode and Plot Input Form Prompts
A14 Error and Status Messages
Error and Status Messages A15
# hex Message 70F
A16 Error and Status Messages
73F
Error and Status Messages A17
75F
A18 Error and Status Messages
78F
Error and Status Messages A19
A20 Error and Status Messages
Advanced Statistics Messages
Error and Status Messages A21
80F
A22 Error and Status Messages
82F
Error and Status Messages A23
85F
A24 Error and Status Messages
87F
Statistics Help Messages
Error and Status Messages A25
HP Solve Application Messages
Time Messages
# hex Message Unit Management
Printing
Programmer’s Doerr
Error and Status Messages A27
A28 Error and Status Messages
System Flags Choose Box Prompts
Error and Status Messages A29
A30 Error and Status Messages
Error and Status Messages A31
A32 Error and Status Messages
Prompts
Error and Status Messages A33
A34 Error and Status Messages
Error and Status Messages A35
Statistics Prompts
Time and Alarm Prompts
A36 Error and Status Messages
Error and Status Messages A37
Symbolic Application Prompts
A38 Error and Status Messages
Error and Status Messages A39
Plot Application Prompts
A40 Error and Status Messages
Error and Status Messages A41
A42 Error and Status Messages
Solve Application Prompts
Error and Status Messages A43
A44 Error and Status Messages
Error and Status Messages A45
CAS Messages
A46 Error and Status Messages
Error and Status Messages A47
DF0A DF0B DF0C DF0D DF0E DF0F
A48 Error and Status Messages
Filer Application Messages
DF1A DF1B DF1C DF1D DF1E DF1F
Error and Status Messages A49
Constants Library Messages
Minehunt Game Prompts
A50 Error and Status Messages
Equation Library Messages
MultipleEquation Solver Messages
Financial Solver Messages
Error and Status Messages A51
Periodic Table Messages
Development Library and Miscellaneous Messages
A52 Error and Status Messages
Units
Tables of Units and Constants B1
B2 Tables of Units and Constants
Tables of Units and Constants B3
B4 Tables of Units and Constants
Tables of Units and Constants B5
Constant Full Name Value in SI Units
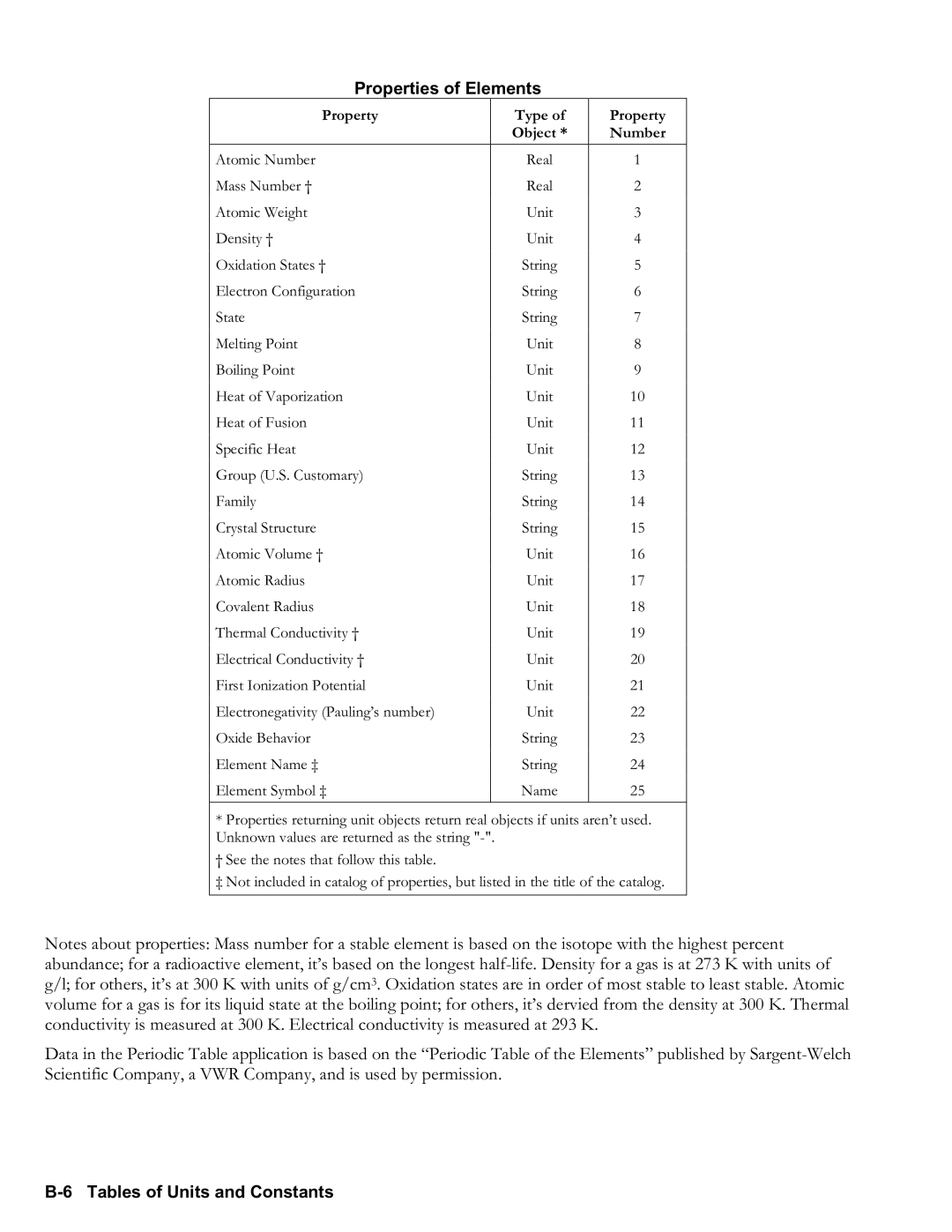
Properties of Elements
B6 Tables of Units and Constants
Not used
System Flags
Flag Description
System Flags C1
C2 System Flags
System Flags C3
C4 System Flags
System Flags C5
C6 System Flags
System Flags C7
Internal use only /0 occurred
C8 System Flags
User Flags
System Flags C9
Page
Reserved Variables D1
Contents of the System Reserved Variables
ΒENTER
Reserved Variables D3
Exited
D4 Reserved Variables
Reserved Variables D5
N1, n2
MHpar
Mpar
Nmines
BAR, etc
Reserved Variables D7
Prtpar
D8 Reserved Variables
S1, s2
Reserved Variables D9
NUMX, Numy
Parameter Description Default Value Command
To use for input into the table
D10 Reserved Variables
Statistical Matrix for Variables 1 to m
Var1 var2 … varm
Reserved Variables D11
EXPFIT, PWRFIT, or Logfit
D12 Reserved Variables
Casdir Reserved Variables
Modulo
D14 Reserved Variables
Object Type Size bytes
Object Size
Technical Reference E1
Pattern Antiderivative
Symbolic Integration
E2 Technical Reference
Technical Reference E3
→DEF Expansions
E4 Technical Reference
Order Operation
Technical Reference E5
International Standard publication No. ISO 31/l197 8 E
E6 Technical Reference
General rules for parallel processing
Group 1 Commands that cannot parallel process
Group 2 Commands that must use Dolist to parallel process
Parallel Processing with Lists F1
Group 4 ADD and +
Group 5 Commands that set modes / states
Group 6 Oneargument, oneresult commands
F2 Parallel Processing with Lists
Group 9 Multipleresult commands
Group 7 Two argument, one result commands
Group 8 Multipleargument, oneresult commands
Group 10 Quirky commands
Using Dolist for Parallel Processing
F4 Parallel Processing with Lists
$ & F
$ & C
$ & D
$ & +
@ & ³ @ & O
Keycode Keystroke Definition
¥ ! &I
~@ & O
@%VARNAME% = varname
~@ & Í @ &†
Keyboard Shortcuts G3
G4 Keyboard Shortcuts
Syntax Example
Menu Numbers
MenuNumber Table H1
Menus 0 through
H2 The MenuNumber Table
MenuNumber Table H3
Menus 118 through
H4 The MenuNumber Table
BuiltIn Library Menus
Menus 178 through
MenuNumber Table H5
Page
Key ALPHARS2 MTH NXT Prob
Command MenuPath Table
+ key
Command Type Library Size Keys Menu First
I2 The Command MenuPath Table
«MODULAR» Addtoreal
MTH List
Arith Modul
CAT Algb
I4 The Command MenuPath Table
PRG NXT
LS&MODE Flag
PRG Test NXT NXT PRG NXT Modes Flag Chinrem
CHR
I6 The Command MenuPath Table
RS&TIME NXT
CAT Delalarm
PRG NXT NXT Time Alrm Delay
PRG NXT NXT Time NXT
I8 The Command MenuPath Table
MTH Matrx NXT
PRG NXT NXT Error Iferr
«POLYNOMIAL» EGV
Else
I10 The Command MenuPath Table
I11
I12 The Command MenuPath Table
PRG Brch NXT
Calc Deriv NXT
«INTEGER»
«DIFF» NXT NXT
I14 The Command MenuPath Table
I15
I16 The Command MenuPath Table
I17
I18 The Command MenuPath Table
I19
I20 The Command MenuPath Table
I21
I22 The Command MenuPath Table
I23
I24 The Command MenuPath Table
I25
I26 The Command MenuPath Table
I27
I28 The Command MenuPath Table
CAT Vtype
Matrices Creat NXT NXT MTH Matrx Make NXT NXT
DUP Menuxy Version
XLIB~
Alphalse
I30 The Command MenuPath Table
SLV NXT
MTH NXT Const «CONSTANTS»
PRG Test «TESTS» NXT
Σline
Σlist
→ALG
I32 The Command MenuPath Table
Character Codes
Code Description
Ascii Character Codes and Translations J1
Code Trans. Description
J2 Ascii Character Codes and Translations
Index
Alpha keyboard
Calculator
59, 531
375 Preserving calculator status
Flags
132
Last argument
Memory
Nested structures
545
Programs
Range 3115
Serial communications
237, D3
Tagged objects
UPs